Principle
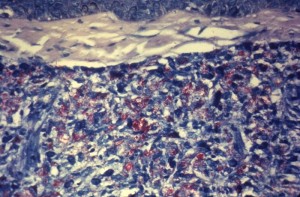
This procedure is used to stain mycobacterium tuberculosis and mycobacterium leprae. These bacteria are also called acid fast bacilli. They stain with carbol fuschin, which is a red dye. They retain the dye when treated with acid, which is because of the presence of mycolic acid in their cell wall.
 Reagents
Reagents
- Carbol fuschin (basic dye)
- Mordant (heat)
- 20% sulphuric acid (decolorizer)
- Methylene blue (counter stain) or Malachite green
Procedure
- Fix the smear of the specimen over the glass slide, either by heating or alcohol fixation.
- Pour carbol fuschin over smear and heat gently until fumes appear. Do not overheat and allow it to stand for 5 minutes, then wash it off with water.
- Pour 20% sulphuric acid, wait for one minute and keep on repeating this step until the slide appears light pink in color. Wash off with water.
- Pour methylene blue, wait for two minutes, again wash with water
- Allow it to air dry and examine under oil immersion lens.
Result
Acid fast bacilli stain pink, straight or slightly curved rods, at times having beaded appearance. The background appears blue due to methylene blue.
Interpretations
If definite bacilli are seen, report as AFB positive. However, it is better to report the result quantitatively as follows:
- > 10 AFB/high power field ->+++
- 1-10 AFB/high power field -> ++
- 10-100 AFB/100 high power fields -> +
- 1-9 AFB/100 high power fields -> exact number
However if no AFB is seen, write the result as ‘no AFB seen’ and never write negative.
Modifications
a. Mycobacterium leprae
Same method is used with 5% sulphuric acid due to less mycolic acid in cell wall.
b. Nocardia and Legionella
1% sulphuric acid is used
c. Stool Specimen
We use 3% acid alcohol instead of sulphuric acid so as to stain Cryptosporidium parvum.
Hot and Cold Ziehl Neelsen Staining
Hot ZN stain is the usual method in which we heat the smear to enhance the dye penetration.
Whereas in cold ZN staining, instead of heating, we increase the concentration of the basic dye and phenol and incorporate a wetting agent chemical.
 howMed Know Yourself
howMed Know Yourself

Thanks i salute you guys. Here there is a rising problem , in ZN staining (AFB) . With consideration of factors that are not inevitable by nature like climate change . The variation of climatic change is the one of the most cause of many bias over in ZN staining of AFB. This is leading to many deaths world wide i therefore request a good set up of the stain at both (National)high level and (district) low level.
thanks very much, i really liked the information on the web site, my concern is that, when we are staining the smear suspecting leprae, what is the concentration of the stains, thanks
Thank you very much for ur contribution in healthy . From Dr . Moses
I am doing the research !
As a medical student .
GRAM STAIN PROCEDURE
Thank you sir , that is very very useful for all lab technician.Kindly inform to anothar microbiological techniq…..
Increasing the carbol fuschin staining time to 25 mins is also used for the Cold ZN staining.
Can I have the actual concentration of the carbol fuschin and example of the wet chemical agent
It is terrific, makes learning easier.
Note.. for mycobacteria Tuberculosis, 3percent acid alcohol is used, while for mycobacteria leprea, 1percent acid alcohol is used.
Very good information
thank u.. So much fr vry detailed info abt z.n method
thank u so much becoz of this
my concept is very much clear thank u once again
Please send the all staining metho and interpretation In my mail address.
Thank you so much for this wonderful information. It really made me to understand the details.
thanks.
Iam a nursing student first yr the results have helped me alot
what about non acid fast?
thanks for the explanation ,it has help me out on my final applied clinical labaratory exam …..thanks again…..
Research about what, would you tell me???
Oh thank u so much this explanation is going to help me alot in my up coming exams under microbes
i do appreciate that really
thank you for infermation
Thanks I got clear and cheaper way of understanding.
oh!! Thank’s you very murch sir on saering this knowlegw.this is a nice idear
Thanks for your short understandable lecture